Introduction: Evolving from Reactive to Predictive Medicine
In today’s precision health landscape, viewing genetic testing and lab markers in isolation is no longer sufficient. Traditional diagnostics like CBC, lipid panels, and CRP have long guided care but are reactive, highlighting dysfunction only after it manifests. In contrast, genetic testing reveals inherited risks early, enabling proactive, personalized interventions. For example, someone with normal lipid labs but carrying APOE ε4 or LDLR variants may still benefit from early cholesterol-lowering strategies. By layering genetic insights with lab markers, healthcare professionals can stratify risk, intervene earlier, and shift from reactive care to predictive, preventive, and personalized medicine crucial for managing chronic disease and optimizing long-term wellness.
The Complementary Role of Genetics and Lab Testing
Genetics: The Blueprint
Genetic data offers insight into an individual’s predisposition to certain traits or conditions. For example:
- MTHFR polymorphisms may reduce methylation efficiency and elevate homocysteine levels.
- TCF7L2 variants increase susceptibility to type 2 diabetes by impairing insulin secretion.
- APOE ε4 alleles are associated with increased risk of Alzheimer’s and dyslipidemia.
These insights act as a predictive map, helping clinicians understand which systems are likely to become dysregulated under stress or poor lifestyle conditions.
Lab Tests: The Real-Time Report Card
On the other hand, blood markers reflect current physiological function:
- A normal fasting glucose level doesn’t rule out future metabolic dysfunction if genetic risk is present.
- A high CRP level indicates ongoing inflammation, but says nothing about genetic capacity to handle oxidative stress.
- Low serum folate may arise due to MTHFR mutations, poor diet, or both, but without genetic insight, the cause remains unclear.
Used in tandem, these tools offer deep visibility into the “what” (lab), the “why” (genetics), and the “when” (timing of intervention).
Case Studies: Real-World Layering for Targeted Interventions
1. Cardiometabolic Risk
- Genetic Report: Variants in FTO and TCF7L2 suggest a predisposition to insulin resistance and obesity.
- Lab Tests: Fasting glucose is normal, but fasting insulin is elevated.
Interpretation: Despite normoglycemia, insulin resistance may be developing. Genetic risk confirms long-term vulnerability.
Intervention: Implement a low-glycemic, anti-inflammatory diet, reduce processed carbohydrate intake, encourage resistance training, and monitor labs every 3–6 months.
2. Methylation and Homocysteine
- Genetic Report: Homozygous MTHFR C677T variant detected.
- Lab Tests: Homocysteine level is 20 μmol/L (elevated), with low serum folate.
Interpretation: Impaired methylation function is active and affecting cardiovascular risk.
Intervention: Recommend methylated folate and B12 supplementation, leafy greens, and homocysteine recheck after 8 weeks.
3. Inflammation and Detoxification
- Genetic Report: Polymorphisms in GSTT1, NQO1, and IL6 indicate poor antioxidant defense and elevated inflammatory response.
- Lab Tests: Elevated CRP and low glutathione levels.
Interpretation: The genetic vulnerability is expressing through systemic inflammation and impaired detox pathways.
Intervention: Implement antioxidant-rich nutrition (e.g., berries, cruciferous vegetables), sulforaphane or NAC supplementation, and stress management techniques.
4. Cognitive Decline Risk
- Genetic Report: APOE ε4 and CLU risk alleles present.
- Lab Tests: Low omega-3 index, high LDL, and elevated homocysteine.
Interpretation: Genetic risk for Alzheimer’s is being amplified by poor lipid and methylation status.
Intervention: Omega-3 supplementation (especially EPA and DHA), methylated B vitamins, Mediterranean diet, cognitive stimulation, and sleep hygiene optimization.
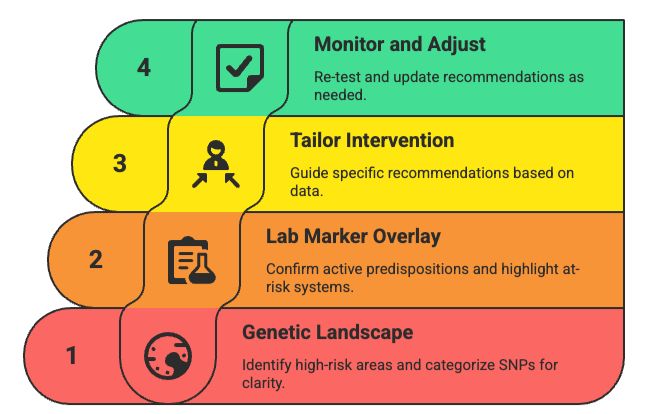
Framework: How to Layer Data Effectively
- Start with the Genetic “Landscape”
- Identify areas of high risk: metabolism, inflammation, neurodegeneration, detox, nutrient transport.
- Categorize SNPs by system or function for clarity.
- Overlay with Current Lab Markers
- Confirm which predispositions are actively expressed.
- Highlight “at-risk but not yet dysfunctional” systems for preventive action.
- Tailor the Intervention Strategy
- Use both data types to guide specific nutrition, supplementation, and lifestyle recommendations.
- Customize frequency of follow-up based on genetic sensitivity and lab trends.
- Monitor and Adjust
- Re-test labs to evaluate efficacy of intervention.
- Update recommendations based on new environmental exposures or life stage changes.
Future of Precision Medicine: From Data to Daily Practice
With wearable devices, AI-powered dashboards, and at-home lab kits, the future of combining genetic testing and lab markers is bright. But technology alone isn’t enoughhealthcare practitioners must develop fluency in interpreting both genomics and biomarkers to shift from illness management to fostering resilience and vitality.
Conclusion
Layering genetic testing and lab markers represents the next frontier in personalized healthcare. This approach doesn’t replace traditional diagnostics, it enhances them, bridging the gap between what is happening now and what may happen next. By embracing this model, physicians, nutritionists, health coaches, and functional medicine practitioners can elevate patient outcomes and redefine proactive, data-informed medicine.
Reference:
- https://doi.org/10.1056/NEJM199902113400607
- https://doi.org/10.3945/jn.114.206599
- https://doi.org/10.1016/S0065-3233(08)60642-7
- https://doi.org/10.1038/ng1732
- https://doi.org/10.1159/000327772
- https://www.sciencedirect.com/science/article/abs/pii/S0002822305021085
- https://doi.org/10.1016/j.ijcard.2016.04.181
- https://doi.org/10.1016/S0140-6736(20)30367-6
- https://doi.org/10.1377/hlthaff.2017.1624
- https://pmc.ncbi.nlm.nih.gov/articles/PMC3547451/
