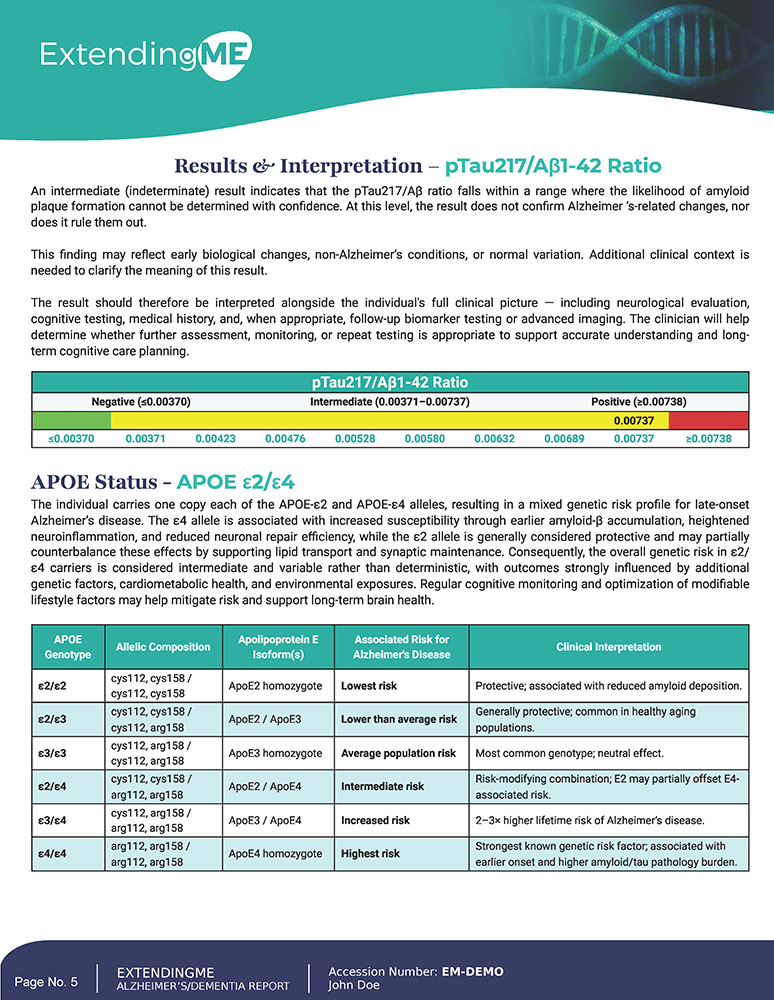
Alzheimer’s Disease Assessment Panel
Integrating Tau–Amyloid and APOE for Precision Cognitive Insight
Overview
The Alzheimer’s Disease Assessment Panel equips clinicians with a clear, integrated view of both current brain pathology and long-term genetic susceptibility to Alzheimer’s disease. By combining an FDA-cleared blood-based biomarker test with APOE genotyping, this panel enables earlier risk awareness, more informed clinical decisions, and personalized brain-health strategies.
Unlike traditional assessments that rely solely on symptoms or family history, this panel delivers objective biological insight helping clinicians identify Alzheimer’s-related changes before cognitive symptoms become clinically obvious.

What This Test Covers
FDA-Cleared Lumipulse® pTau217 /
Aβ1-42 Plasma Ratio Test
This panel includes the FDA-cleared Lumipulse® blood test, offering a less invasive and more accessible alternative to PET imaging or cerebrospinal fluid analysis.
It evaluates two critical Alzheimer’s-associated biomarkers:
- pTau217
- β-Amyloid 1-42 (Aβ1-42)
The pTau217 / Aβ1-42 ratio provides insight into early amyloid-related brain changes, often preceding noticeable memory loss or cognitive decline. This allows clinicians professionals to identify biological risk earlier in the disease continuum.
Health Support Insights
- Cognitive Exercise Training Evidence-informed activities that support neuroplasticity, memory, and long-term brain resilience aligned with the individual’s overall risk profile.
- Nutritional Insights Targeted nutrition insights to support amyloid clearance, mitochondrial health, and inflammation balance.
- Lifestyle Strategies Key modifiable factors including sleep, stress, metabolic health, and cardiovascular function.
- Supplement Insights Evidence-based supplement insights aligned with individual biological and genetic context.
- Labs to Consider Select laboratory markers to support clinical decision-making and further personalize nutrition, lifestyle, and supplement strategies.
APOE Genotype Analysis
The report includes APOE genotyping, the most extensively studied genetic factor in late-onset Alzheimer’s disease.
● APOE-ε4
● APOE-ε2
● APOE-ε3
This genetic layer provides context for lifetime susceptibility, helping clinicians tailor risk communication and preventive strategies.
Integrated Risk Interpretation -
Biological Activity + Genetic Predisposition
By uniting current biomarker activity with genetic risk, this panel delivers a more complete and clinically meaningful Alzheimer’s risk profile. Clinicians gain clarity on whether changes reflect active pathology, inherited vulnerability, or both, enabling smarter monitoring and earlier action.


Why It Matters
Alzheimer’s disease begins years, often decades before diagnosis. Relying on symptoms alone delays intervention.
This panel allows clinicians to
- Identify early Alzheimer’s-related biological changes
- Move from reactive care to preventive brain health strategies
- Support informed, confident patient conversations
- Offer personalized guidance backed by objective data
Who Should Take This Test
This panel is designed for
Physicians and neurologists
Physicians and neurologists
Preventive and longevity-focused clinicians
Functional and integrative medicine practitioners
Clinicians managing patients with memory concerns or family history of dementia
Ideal for practices aiming to deliver proactive, precision-driven cognitive health care
What You’ll Receive
Comprehensive Report
• FDA-cleared pTau217 / Aβ1-42 blood biomarker results
• APOE genotype analysis with clear interpretation
• Integrated Alzheimer’s risk overview

Personalized Insights
Clinically relevant insights across cognition, nutrition, lifestyle, supplements and labs to consider.

Provider Portal Access
Secure digital access to the ExtendingME Provider Portal for test ordering and result tracking.

Turnaround Time
Results will be available approximately 2 weeks from the receipt of the sample and will be securely delivered through the Provider Portal, allowing efficient review and patient follow-up.
Collection Method
ExtendingME kits include all necessary self-collection devices for non-invasive, at-home saliva collection, making it easy to deliver real-time, data-driven healthcare solutions without the need for lab visits.

